 soft ferromagnet of second-order phase transition, abrupt phase change at T C, the resultant large magnetic entropy change of 4.4 J kg −1 K −1 and temperature averaged entropy change of 3.914 J kg −1 K −1 (10 K) at the low magnetic field of 1.5 T are expected to facilitate application prospects as a magnetic refrigerants for room temperature working range. The magnetic interactions are governed by the Tri-Critical Mean Field model with emphasis on the coexistence of long-range and short-range interactions. What is Entropy Change Since entropy is a state function, the entropy change of a system depends only on the initial and final state irrespective of the path taken. 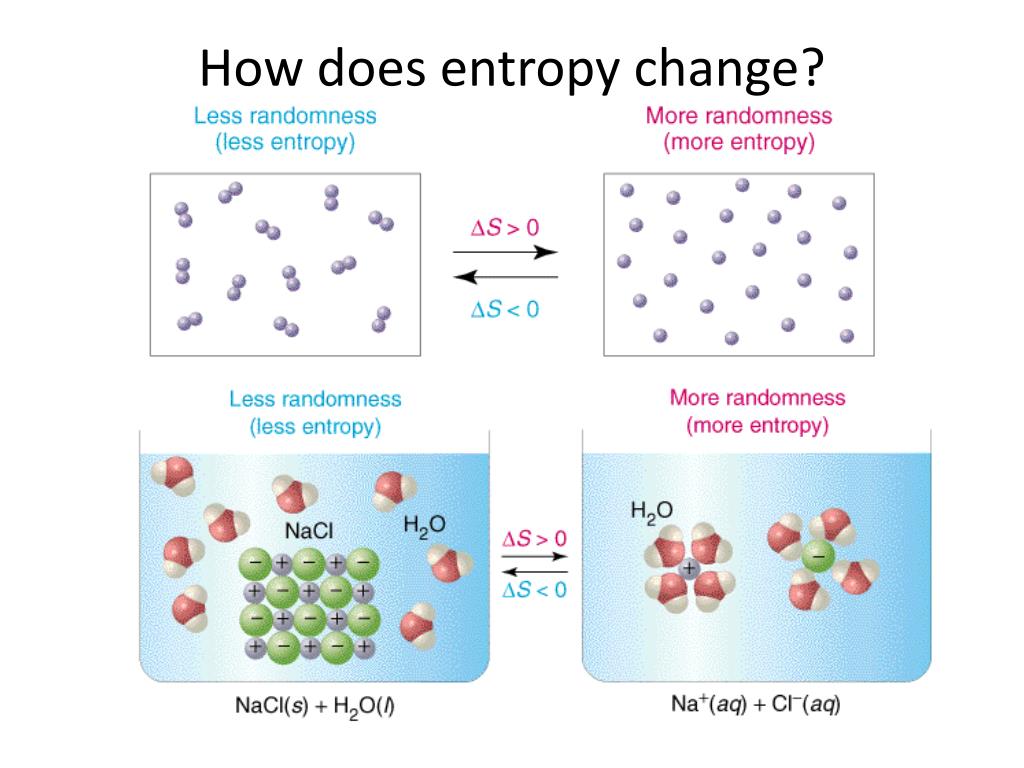
It is accompanied by an increase in the disorder of the water molecules. The entropy change of a system may be defined as the integral of all the terms involving heat exchanged (q) divided by the absolute temperature (T) during each. Calculate the change in entropy when 10 kg of air is heated at constant volume from a pressure of 101325 N/m2 and a temperature of 20oC to a pressure of 405300. As there is a change in the state from solid. This is a significant increase in entropy, because it takes place at a relatively low temperature. Hence, it is found that there are entropy changes during the phase transition from solid to gaseous and vice versa. One mole of a solid is produced from two moles of ions in. The change in entropy is positive, because heat transfers energy into the ice to cause the phase change. Nearly 100–500 percent increases in magnetic entropy change was realised over existing parent compounds and the entropy changes are greater than the prototype, Gd of active magnetic refrigeration. Since gases have so much more entropy than solids, the entropy change of this reaction is positive. So we can calculate the change in entropy of the hot reservoir for a hypothetical reversible process in which 4000 J of heat transfer occurs from it then we do. Phenomenal improvement in magnetic sensitivity at low magnetic fields was achieved with T C close to room temperature. The sample has a soft ferromagnetic nature with a single magnetic transition occurring at 275 K. 
The chemical additive Bi 2O 3 has assisted in grain growth to form densified specimens even at low sintering temperatures. Unlike enthalpy or internal energy, it is possible to obtain absolute entropy values by measuring the entropy change that occurs between the reference point of 0 K corresponding to S 0 J/ (molK) and 298 K. A 10% Bi ion substitution at the La site has led to the relatively higher symmetry R c rhombohedral structure showing improvement in Mn–O–Mn bond angles. The bismuth (Bi)-substituted La 0.85− xBi xNa 0.15MnO 3 ( x = 0 and x = 0.1) manganites have been investigated for achieving room temperature magnetic entropy change at low magnetizing intensities. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
